David Chaupis-Meza
César Cárcamo
King Holmes
Patricia García
BRIEF REPORT
Population
seroprevalence of celiac disease in urban areas of Peru
Katherine Baldera ![]() 1,
Licensed in Medical Technology with a specialty in Clinical
Laboratory
1,
Licensed in Medical Technology with a specialty in Clinical
Laboratory
David Chaupis-Meza ![]() 1,
bachelor of Science in Medical
Technology
1,
bachelor of Science in Medical
Technology
César Cárcamo ![]() 2, medical doctor, PhD in epidemiology
2, medical doctor, PhD in epidemiology
King Holmes ![]() 3, medical doctor, PhD in Microbiology
3, medical doctor, PhD in Microbiology
Patricia García ![]() 2,
medical doctor, PhD in Medicine
2,
medical doctor, PhD in Medicine
1
Escuela
de Tecnología Médica de la Facultad de Medicina, Universidad Peruana Cayetano
Heredia, Lima, Peru.
2 Unidad
de Epidemiología, ETS y VIH de la Facultad de Salud Pública y Administración,
Universidad Peruana Cayetano Heredia, Lima, Peru.
3 School
of Public Health and Community Medicine, University of Washington, Seattle,
Washington, EE. UU.
ABSTRACT
The
objective of the study was to determine the seroprevalence of celiac disease
(CD) in urban areas of Peru using a population-based sample. A random sample of
women and men 18 to 29 years old from 26 cities in Peru was screened. An
anti-tissue transglutaminase IgA kit was used for the detection of CD. Results
higher than 20 AU / ml were considered positive. The
weighted prevalence of celiac disease was 1.2% (CI 95%: 0.0% - 2.4%), thus the
estimated number of people living with CD in Peru was 341,783. CD prevalence in
Peru is similar to the world average.
Keywords: Celiac Disease; Seroprevalence; Seroepidemiologic Studies; Transglutaminases (source: MeSH NLM).
INTRODUCTION
Intolerance
to gluten (whether from wheat, barley or rye) leads to a wide spectrum of
chronic enteropathies: a) gluten allergy; b) non-celiac sensitivity to gluten
(NCSG); and c) celiac disease (CD). CD is an autoimmune disorder with a genetic
predisposition linked to the alleles HLA-DQ2 and HLA-DQ8 (1,2).
Despite its recognized characterization, its diagnosis is always elusive.
When compared to CD, NCSG shows no association with any genetic or
immunological alteration, nonetheless both conditions present a similar
clinical picture. This seems to be due to the fact that a gluten-derived
fragment, alpha-gliadin, would be the main target epitope associated with
duodenal disorders present in both CD and NCSG (3).
Depending on its clinical presentation, CD can present evident
symptoms (diarrhea, steatorrhea, fatigue, dyspepsia, and malnutrition), an
extragastrointestinal clinical picture (iron deficiency anemia, fatigue,
depression, peripheral neuropathies, and cerebellar ataxia), or an asymptomatic
picture
(4), often detected by routine serological screening or by its
association with chronic complications such as malignant lymphomas (5).
Worldwide, the prevalence of CD is 1%, with a female:male ratio of 2.8:1. It is assumed that Latin America and
Europe have similar prevalence of CD, which may vary between 0.46 and 0.64% (6).There
are no population-based studies of prevalence in Peru, but there are clinical
cases of CD that warn of the need for national serological mapping of the
disease (7,8,9).
|
KEY MESSAGES |
|
Motivation for the study:
Celiac disease has different clinical presentations, being the undiagnosed
cases the larger group yet to be recognized. A serological screening can be
an early detection test for celiac disease; furthermore, it would avoid fatal
complications.
Main findings:
This is the first
“extra-nosocomial” population-based study in Peru. A prevalence of celiac
disease of 1.2% was found using the IgA tissue antitransglutaminase test.
Implications:
Further population-based studies of celiac disease in Peru are recommended. |
These tests
detect antibodies that are reactive to gluten (IgA and/or IgG), such as
anti-endomysial antibody (EMA), anti-glia-din antibody (AGA), and anti-
transglutaminase antibody (tTGA). The latter is used for the early detection of
CD at any age, which is then confirmed by an intestinal biopsy (10).
The purpose of the study was to determine the seroprevalence of CD in urban
populations of Peru using a population-based sample.
THE STUDY
Subjects and study population
The study uses demographic and biological sampling information
from the PREVEN study (11), which included women and men between
18 and 29 years old in 26 cities in Peru, selected through a multi-stage
cluster sampling in households located in urban areas. Our study selected 1,208
samples by simple random sampling from 17,293 samples collected between 2005
and 2007 by the PREVEN study. Participants completed an epidemiological
questionnaire and provided biological samples. The serum obtained was stored at
–20 °C in the serum bank of the Research and Development Laboratories (LID) of
Universidad Peruana Cayetano Heredia (UPCH).
Sample processing
Hemolyzed,
lipemic samples were discarded; as well as cryovials with insufficient
remaining volume (<10ul) of serum or samples without epidemiological
information. The samples were processed by colorimetric enzyme-linked
immunosorbent assay (ELISA). The IgA tissue antitransglutaminase kit (Diametra
Diagnostic, Italy) was used according to the manufacturer’s protocol, which has
a sensitivity of 93% and specificity of 95% (12). The samples and the reagent
kit were defrosted at room temperature for about 30 minutes. One positive and
one negative control were included. The samples were diluted 1:10 and placed in
microwells coated with recombinant tissue transglutaminase. They were incubated
for 30 minutes at room temperature.
The supernatant was discarded and the microwells were washed
three times to remove the remaining unbound components. Anti-human IgA
conjugate marked with horseradish peroxidase (HRP) was then added and incubated
for 30 minutes at room temperature. After washing, the excess conjugate was removed,
the substrate was added with trimethylbenzidine (TMB) and incubated for 15
minutes at room temperature protected from the light. The reaction was stopped
with sulphuric acid.
The sample
reading was made with a 450 nm filter and a 650 nm reference filter. As a
confirmatory test, those samples with results >20 AU/ml were subjected to a
second ELISA following the same procedures described. Those samples in which
the second ELISA showed values >20 AU/ml were considered positive. Samples
with ≤20 AU/ml values in the first or second ELISA were considered negative.
Statistical Analysis
For each
sample included in the study, an expansion factor was calculated, corresponding
to the inverse of the probability of participation. The probability of
participation was the product of several probabilities: the probability of
selection of the cluster within the city, the probability of selection of the
dwelling within the cluster, the probability of selection of the participant
within the dwelling, and the probability of selection of the individual for
participation in the present study. Estimates were also adjusted for city level
stratified sampling. Weighted prevalence is presented, with its respective
confidence intervals. Pearson’s chi-square test for weighted samples was used
for the comparison of proportions. Stata 8.2 (College Station, Texas) was used
for all calculations.
Ethical aspects
Participants provided verbal consent for their participation in
the study. The original study’s informed consent form contemplated the storage
of samples for future studies without the inclusion of personal identifiers. As
part of this study, only codes were used to identify the samples and link them
to their epidemiological information. The present study was approved by the
Institutional Research Ethics Committee of Universidad Peruana Cayetano Heredia
(code SIDISI 60226).
RESULTS
Out
of the 1,208 selected samples, 107 were discarded for being hemolyzed (73),
lipemic (1), insufficient volume (18) and lack of epidemiological information
(15). Finally, 1,101 samples were incl44uded in the analysis, of which 420 were
male and 681 were female (Table 1).
Table 1. Demographic characteristics of the 1,101
participants selected
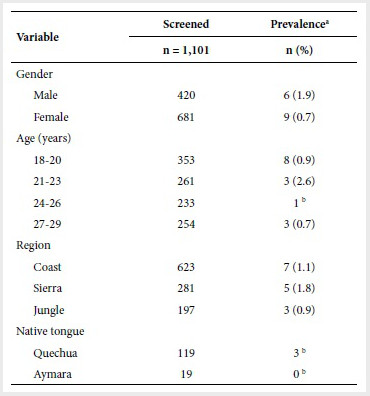
a Weighted prevalence; b could not
be calculated as there was only one
A weighted prevalence of CD of 1.2% (95% CI 0.0-2.4) was
obtained. The sample is representative of 3,399,734
people aged 18-29 years, living in urban areas in Peru. Thus, it is estimated
that in this population there are 40 797 people with CD (95% CI: 0-81 594).
Although the weighted prevalence in women (0.7%) was lower than
in men (1.9%), this difference is not statistically significant (p=0.253).
Similarly, the prevalence in the 21-23 age group is higher than for other age
groups, no statistically significant difference is found (p=0.144).
Of the total number of individuals, 623 lived in the coastal
region, 281 to the sierra and 197 to the jungle. The prevalence of CD in the
sierra (1.8%) was not significantly higher than that found on the coast or in
the jungle (1.1 and 0.9%, respectively).
Although
few participants spoke Quechua or Aymara (119 and 19, respectively), no
statistically significant differences were found in the prevalence of CD
between these and the rest of the participants in the study (p=0.288 and
p=0.811, respectively).
DISCUSSION
This
is the first study in Latin America to evaluate the prevalence of CD by
serological screening for tissue antitransglutaminase IgA in a population-based
sample, and a prevalence of 1.2% was found. If the prevalence for other ages
and geographical areas of the country in 2007 had been the same, then it is estimated
that for that year the number of people living with celiac disease in Peru was
341,783.
In the United States, the prevalence of undiagnosed CD is
increasing. A study determined the presence of IgA tissue antitransglutaminase
in stored samples (for at least 20 years at –20 °C), to determine changes in
prevalence over a 50-year period (13). Samples taken between 1948
and 1954 showed a prevalence of 0.2% and those taken between 1995 and 2003
showed a prevalence of 0.8%, both in adults over 50 years. On the other hand,
in samples of persons aged 18 to 49 taken between 2006 and 2008, a prevalence
of 0.9% was found. Previously in the mentioned study the presence of IgA was
revealed in the old samples using nephelometry.
Another study conducted in Argentina (14), using AGA (IgA and
IgG) and EmA (IgA) as serological markers, determined a CD prevalence of 0.6%.
Our study reveals a higher prevalence, probably because of the age group and
the type of serological test used. On the other hand, a study in Brazil (15) in people of 18-65 years old without anemia
found a prevalence of 0.33% using the anti-TBM IgA and anti-EmA IgA test. The
low prevalence found may respond to the fact that anemia is an atypical
presentation of CD.
Epidemiological studies exploring the population risk of
presenting CD are scarce in Latin America (6). In Peru, the few clinical
reports that exist warn of the presence of CD in a “low frequency” and linked
to the classic type of the disease (7-9,16).
Serological screening for CD is recommended instead of the use of
invasive methods such as biopsies, being an early diagnostic choice to rule out
CD. According to ESPGHAN (European Society for Pediatric Gastroenterology,
Hepatology and Nutrition) (17), the diagnosis of CD can be confirmed
with anti-transglutaminase levels above ten times the upper normal limits (≥20
U/ml, reference value for our study), this levels are compared to Marsh-3 type
villous atrophy. This emphasizes the usefulness of the IgA tTGA serological
test as the most reliable serological marker for CD, not only because of its
high specificity and sensitivity, but also because of its usefulness, as
samples can be stored for extended periods of time,being
possible to reuse them in cryopreserved samples (18), as in our case.
Indeed, having a reliable serological test would allow for a reduction in costs
and time in the diagnosis of CD.
The absence of information on gastrointestinal symptoms and
history of CD could constitute a limitation of the present study, however, the
clinical correlation of serology is quite well known.
Our study found a higher prevalence of CD in people from the
sierra. This could correspond to genetic differences, or to environmental
factors, such as height and consequent hypoxia. Although other studies have
found higher prevalence in women (6), our study found higher
prevalence in the male population, a difference for which we found no cause.
In
conclusion, the present study is the first to be carried out in Peru based on a
population sample of young adults, showing a higher prevalence than reported in
other studies of the American continent, but similar to the world average. More
studies of seroprevalence in populations that are atypical for CD are
suggested, taking into account the different forms of clinical presentation,
since in most cases celiac disease has no apparent symptoms.
REFERENCES
1. Sapone A, Bai JC, Ciacci C,
Dolinsek J, Green PHR, Hadjivassiliou M, et al. Spectrum of
gluten-related disorders: consensus on new nomenclature and classification. BMC
Med. 2012;10:13.
2. Elli L, Branchi F, Tomba C,
Villalta D, Norsa L, Ferretti F, et al. Diagnosis of gluten related
disorders: Celiac disease, wheat allergy and non-celiac gluten sensitivity.
World J Gastroenterol. 2015;21(23):7110–9.
3. Lebwohl B, Ludvigsson JF,
Green PHR. Celiac disease and non-celiac gluten sensitivity. BMJ. 2015;351:h4347.
4. Rashid M, Lee J. Serologic
testing in celiac disease: Practical guide for clinicians. Can Fam Physician
Med Fam Can. 2016;62(1):38–43.
5. Kelly CP, Bai JC, Liu E,
Leffler DA. Advances in diagnosis and management of celiac disease.
Gastroenterology. 2015;148(6):1175–86.
6. Parra-Medina R,
Molano-Gonzalez N, Rojas-Villarraga A, Agmon-Levin N, Arango M-T, Shoenfeld Y, et
al. Prevalence of celiac disease in latin america: a systematic review and
meta-regression. PloS One. 2015;10(5):e0124040.
7. Vera A, Frisancho O, Yábar
A, Carrasco W. Enfermedad Celiaca y Obstrucción Intestinal por Linfoma de
Células T. Rev Gastroenterol Peru. 2011;31(3):278–81.
8. Llanos O, Matzumura M,
Tagle M, Huerta-Mercado J, Cedrón H, Scavino J, et al. Enfermedad
celiaca: estudio descriptivo en la Clínica Anglo Americana. Rev Gastroenterol
Peru. 2012;32(2):134–40.
9. Tagle M, Nolte C, Luna E,
Scavino Y. Coexistencia de Enfermedad Celíaca y Hepatitis Autoinmune. Reporte
de un caso y revisión de la literature. Rev Gastroenterol Peru. 2006;26(1):80–3.
10. Rostom A, Murray JA,
Kagnoff MF. Medical Position Statement on Celiac Disease. Gastroenterology.
2006;131(6):1977–80.
11. García PJ, Holmes KK,
Cárcamo CP, Garnett GP, Hughes JP, Campos PE, et al. Prevention of
sexually transmitted infections in urban communities (Peru PREVEN): a
multicomponent community-randomised controlled trial. Lancet. 2012;379(9821):1120–8.
12. Kowalski K, Mulak A,
Jasinska M, Paradowski Leszek. Diagnostic challenges in celiac disease. Adv
Clin Exp Med. 2017; 26(4): 729–37.
13. Rubio-Tapia A, Ludvigsson
JF, Choung RS, Brantner TL, Rajkumar SV, Landgren O, et al. Increased
mortality among men aged 50 years old or above with elevated IgA
anti-transglutaminase antibodies: NHANES III. BMC Gastroenterol. 2016;16(1):136.
14. Gomez JC, Selvaggio GS,
Viola M, Pizarro B, la Motta G, de Barrio S, et al. Prevalence of celiac
disease in Argentina: screening of an adult population in the La Plata area. Am
J Gastroenterol. 2001;96(9):2700–4.
15. Melo SBC, Fernandes MIM,
Peres LC, Troncon LEA, Galvão LC. Prevalence and demographic characteristics of
celiac disease among blood donors in Ribeirão Preto, State of São Paulo,
Brazil. Dig Dis Sci. 2006;51(5):1020–5.
16. Arévalo F, Roe E,
Arias-Stella-Castillo J, Cárdenas J, Montes P, Monge E. Low serological
positivity in patients with histology compatible with celiac disease in Perú.
Rev Esp Enferm Dig. 2010;102(6):372–5.
17. Smarrazzo A, Misak Z,
Costa S, Mičetić-Turk D, Abu-Zekry M, Kansu A, et al. Diagnosis of
celiac disease and applicability of ESPGHAN guidelines in Mediterranean
countries: a real life prospective study. BMC Gastroenterol. 2017;17(1):17.
18. Wengrower D, Doron D,
Goldin E, Granot E. Should stored Serum of Patients Previously Tested for
Celiac Disease Serology be Retested for Transglutaminase Antibodies?. J Clin Gastroenterol. 2006;40(9):806-8.
Funding:
Fundación Instituto Hipólito Unanue.
Citation:
Baldera K, Chaupis-Meza D,
Cárcamo C, Holmes K, García P. Seroprevalencia poblacional de la enfermedad
celíaca en zonas urbanas del Perú. Rev Peru Med Exp Salud Publica.
2020;37(1):63-6. Doi: https://doi. org/10.17843/rpmesp.2020.371.4507.
Correspondence to:
David
Chaupis Meza; Universidad Peruana Cayetano Heredia; Av. Honorio Delgado 430,
Lima 31, Peru;
david.chaupis.m@upch.pe.
Authorship
contribution: KB participated in the technical execution,
data analysis and obtained the funding for the study. DCM participated in the
conception and design of the study, interpretation of the results and writing
of the first version of the manuscript. CC participated in the statistical analysis
and interpretation, critical review and writing of the final version of the
article. Both KH and PG are the principal investigators of the PREVEN study.
All the authors finally approved the last version of the article.
Conflicts
of interest:
All authors have none to declare.
02/05/2019
Approved:
29/01/2020
Online:
23/03/2020